The Promise of In-Womb Treatment: A New Frontier in Treating Genetic Disorders Before Birth
The concept of treating diseases before birth might sound like something out of science fiction, but recent advances in genetic testing and molecular medicine are turning this dream into reality. Imagine diagnosing a genetic disorder during pregnancy and, instead of waiting for the baby to be born, intervening while the fetus is still developing. This is not just a hopeful possibility but a tangible approach that could change the way we treat genetic diseases.
Early Diagnosis and Intervention: A Game Changer
For years, genetic diseases have been diagnosed early during pregnancy, but the ability to do something about them, until now, was limited. Early intervention for disorders such as thalassemia or cystic fibrosis has long been a challenge, often because these conditions manifest after birth when the damage has already been done. However, thanks to recent innovations in non-invasive genetic testing, it is now possible to diagnose conditions like these as early as the first trimester, providing a critical window for intervention before birth.
Despite these advances, many single-gene disorders continue to result in significant morbidity and mortality in neonates and children. But what if we could intervene while the child is still in the womb, correcting or treating the genetic defect before it leads to irreversible damage? That idea has gained ground recently, and one remarkable study published in the New England Journal of Medicine this February has provided some evidence that in-utero treatment can indeed be a life-saving option.
Spinal Muscular Atrophy (SMA): First genetic disorder treated in the Womb
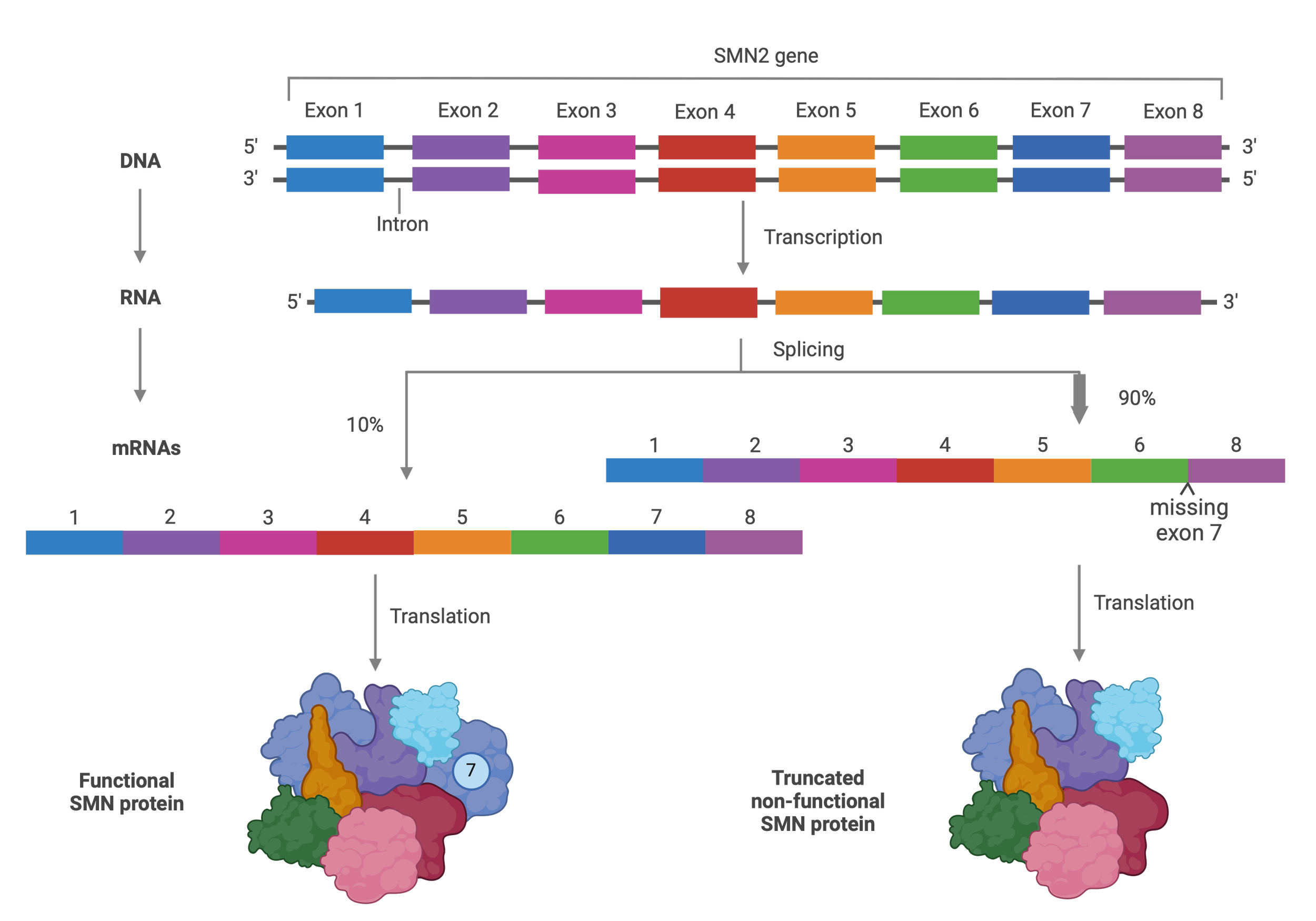
Spinal Muscular Atrophy (SMA) is a rare but severe genetic disorder caused by a lack of the SMN protein, which is essential for motor neuron survival. In healthy individuals, two genes carry the instructions to produce this protein: SMN1, responsible for approximately 80% of SMN protein production, and SMN2, which contributes around 20% due to an RNA splicing error that leads to an incomplete and mostly non-functional protein—the error in processing excludes the exon 7 of the SMN2 gene.
In 94% of SMA cases, patients lack a functional SMN1 gene, leaving them reliant on the insufficient SMN protein produced by the gene SMN2. The severity of SMA correlates with the number of SMN2 gene copies a patient has—more copies mean a greater ability to compensate for the missing SMN1 gene.
The lack of SMN protein in SMA leads to the degeneration of motor neurons in the spinal cord, resulting in muscle weakness, difficulty breathing, and ultimately, respiratory failure. In its most severe form, SMA Type 0, symptoms begin even before birth, leading to an extremely poor prognosis, with infants rarely surviving beyond six months.
The central dogma of molecular biology & RNA splicing explained:
Genetic information (DNA) is used in our cells to produce proteins, which carry out essential functions. DNA acts as a vast library and contains many books (genes), each gene is copied into an mRNA molecule that serves as a concise summary of a specific book or gene, and ribosomes use this information to assemble a protein. This process ensures that cells function properly and that proteins like SMN are produced as needed.
But before the ribosomes make the protein the mRNA molecule is processed, that’s called RNA splicing, a cellular process that removes non-coding segments (introns) from a gene transcript, leaving only the essential coding parts (exons). In the case of the SMN2 gene, a crucial exon (exon 7) is often skipped, leading to a truncated and non-functional protein. This splicing error is the key issue in SMA, and treatments aim to correct it.
The Role of Risdiplam: A New Hope for SMA
While there is no cure for SMA, understanding its molecular mechanisms has enabled the development of targeted therapies. One such breakthrough is risdiplam, a small-molecule drug designed to modify RNA splicing and increase the production of full-length SMN protein from the SMN2 gene. By stabilizing exon 7 inclusion, risdiplam enhances SMN2’s ability to produce functional protein, offering a promising treatment option.
Typically, risdiplam is administered postnatally, with better outcomes observed the earlier it is given. However, a groundbreaking case study has now demonstrated the feasibility of administering risdiplam before birth.
A recent study at St. Jude Children's Research Hospital documented the first-ever in-utero administration of risdiplam. A pregnant mother carrying a fetus diagnosed with SMA Type 1 received oral risdiplam during the last seven weeks of pregnancy. Upon birth, the baby displayed no signs of SMA—showing normal muscle tone, movement, and breathing function. Now, at two and a half years old, the child continues risdiplam therapy and remains free of SMA symptoms.
This case provides compelling evidence that early intervention—before the onset of symptoms—could alter the course of SMA, potentially preventing neuronal damage before it occurs. Beyond this individual success, the study raises broader questions about how prenatal treatments might reshape clinical protocols for genetic disorders. Could early intervention become a standard of care for diseases where damage begins before birth? What regulatory and ethical considerations must be addressed to make these treatments more widely available?
The Future of Prenatal Gene Therapy
Current treatments like risdiplam aim to manage genetic disorders by improving symptoms but do not fix the underlying genetic mutation. Gene therapy, in contrast, seeks to correct the genetic defect itself, offering a potential cure.
In utero gene therapy has primarily relied on stem-cell transplantation and viral-mediated gene delivery, which, while promising, come with significant limitations. Stem-cell transplantation involves introducing healthy stem cells into the fetus to replace genetically damaged cells. These stem cells have the potential to develop into different types of cells needed by the body. However, ensuring that the transplanted cells integrate correctly and function as intended remains a major challenge. Viral-mediated gene delivery uses modified viruses to carry healthy copies of a gene into fetal cells. The virus acts like a delivery vehicle, inserting the corrected genetic material into the cells' DNA. While this method has been effective in some treatments, it comes with risks, such as immune system reactions or the inserted gene integrating in the wrong place, which could cause unintended effects.
Recent research has demonstrated that nanoparticle-based gene editing allows for gene correction in its endogenous environment. This technique involves using ultra-small particles to transport DNA fragments directly into fetal cells, allowing for precise corrections to genetic mutations without relying on viral vectors. This approach minimizes immune reactions and the risk of unintended genetic changes, making it a promising alternative for prenatal interventions.
β-thalassemia is a genetic blood disorder that reduces the production of hemoglobin, the molecule in red blood cells that carries oxygen throughout the body. This condition can lead to severe anemia, requiring frequent blood transfusions. A study on β-thalassemia in mice showed that intravenously or intra-amniotically administered nanoparticles containing peptide nucleic acids (PNAs) and donor DNA fragments could correct a disease-causing mutation. This resulted in sustained postnatal elevation of hemoglobin levels, improved red blood cell morphology, and increased survival—highlighting the potential of non-viral, site-specific genome editing for treating genetic disorders in the womb. These findings suggest that in-utero gene therapy could become a transformative strategy for preventing severe genetic conditions. However, significant hurdles remain, including optimizing delivery methods, ensuring safety across various developmental stages, and addressing ethical concerns regarding the modification of fetal DNA—even if doctors aren’t creating “designer babies”.
Conclusion: A New Era in Fetal Medicine
Early genetic testing and in-utero treatments offer transformative possibilities for genetic disorders. The groundbreaking risdiplam study marks an important milestone, demonstrating that understanding the molecular mechanisms of disease and intervening before birth can dramatically improve outcomes. While prenatal gene therapy remains in its early stages, the potential to correct genetic conditions before they cause irreversible harm is becoming increasingly viable. This research is just the beginning, and future studies will determine whether these treatments can become routine clinical practice.
What does this mean for the future of medicine? Will we soon see prenatal therapies for conditions like sickle cell disease, Duchenne muscular dystrophy, or even certain forms of cancer predisposition? Only time and continued research will tell—but one thing is clear: the future of medicine is unfolding inside the womb.
Would you like to stay updated on the latest breakthroughs in biomedical science?
Subscribe to my blog and join me in exploring the next frontier of medicine!